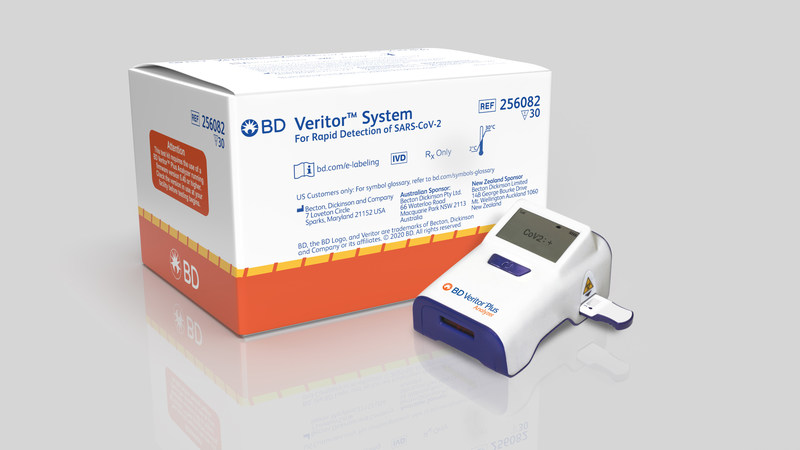
The bd veritor system for rapid detection of sars cov 2 is used for patients suspected of having covid 19 by their healthcare provider and is a simple to administer point of care antigen test that delivers reliable results in 15 minutes.
Bd veritor system for rapid detection of sars cov 2 cost.
Learn about the bd veritor system for the rapid detection of sars cov 2 with the bd veritor plus analyzer.
Bd veritor system for rapid detection of respiratory syncytial virus rsv package insert 8086098 2017 08.
The bd veritor system for rapid detection of sars cov 2 is a chromatographic immunoassay for the direct and qualitative detection of sars cov 2 antigens in nasal swabs from patients with signs and symptoms who are suspected of covid 19.
Demonstrated that the test is capable of.
Becton dickinson and company.
The bd veritor plus system for rapid detection of sars cov 2 assay is deemed to be a clia waived immunoassay designed to be used in health care settings to provide an aid to rapid diagnosis of covid 19 in symptomatic individuals.
Bd statement on bd veritor plus system for rapid detection of sars cov 2 assay franklin lakes n j.
Bd veritor system for rapid detection of flu a b clia waived kit package insert 8087667 2018 06.
15 2020 bd becton dickinson and company nyse.